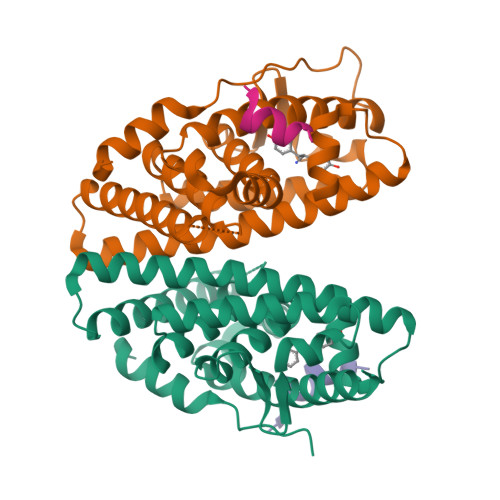
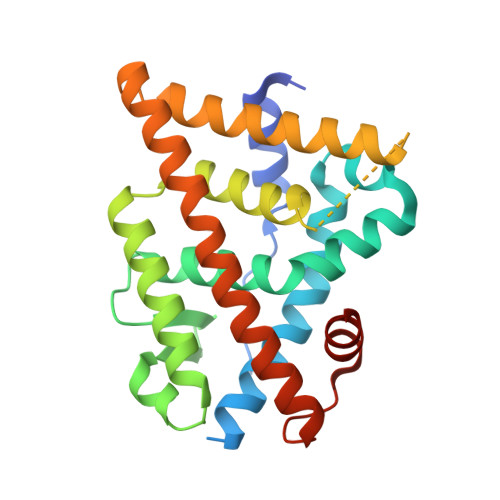
Evaluating the correlation of binding affinities between isothermal titration calorimetry and fragment molecular orbital method of estrogen receptor beta with diarylpropionitrile (DPN) or DPN derivatives.
Handa, C., Yamazaki, Y., Yonekubo, S., Furuya, N., Momose, T., Ozawa, T., Furuishi, T., Fukuzawa, K., Yonemochi, E.(2022) J Steroid Biochem Mol Biol 222: 106152-106152
- PubMed: 35810932
- DOI: https://doi.org/10.1016/j.jsbmb.2022.106152
- Primary Citation of Related Structures:
7XVY, 7XVZ, 7XWP, 7XWQ, 7XWR - PubMed Abstract:
Estrogen receptors (ERs) are ligand-activated transcription factors, with two subtypes ERα and ERβ. The endogenous ligand of ERs is the common 17β-estradiol, and the ligand-binding pocket of ERα and ERβ is very similar. Nevertheless, some ERβ-selective agonist ligands have been reported. DPN (diarylpropionitrile) is a widely used ERβ-selective agonist; however, the structure of the ERβ-DPN complex has not been solved. Therefore, the bound-state conformation of DPN and its enantioselectivity remain unresolved. In this report, we present the structures of the complexes of ERβ with DPN or its derivatives that include a chlorine atom by the X-ray crystallography. Additionally, we measured the binding affinity between ERβ and DPN or derivatives by isothermal titration calorimetry (ITC) and estimated the binding affinity by fragment molecular orbital (FMO) calculations. We also examined the correlation between the ITC data and results from the FMO calculations. FMO calculations showed that S-DPN interacts strongly with three amino acids (Glu305, Phe356, and His475) of ERβ, and ITC measurements confirmed that the chlorine atom of the DPN derivatives enhances binding affinity. The enthalpy change by ITC correlated strongly with the interaction energy (total IFIEs; inter-fragment interaction energies) calculated by FMO (R = 0.870). We propose that FMO calculations are a valuable approach for enhancing enthalpy contributions in drug design, and its scope of applications includes halogen atoms such as chlorine. This study is the first quantitative comparison of thermodynamic parameters obtained from ITC measurements and FMO calculations, providing new insights for future precise drug design.
Organizational Affiliation:
Central Research Laboratory, Kissei Pharmaceutical Co., Ltd., 4365-1, Hotakakashiwabara, Azumino, Nagano 399-8304, Japan; School of Pharmacy and Pharmaceutical Sciences, Hoshi University, 2-4-41 Shinagawa, Tokyo 142-8501, Japan. Electronic address: chiaki_handa@pharm.kissei.co.jp.